
Monarch Butterfly frozen but not dead.
Across the biosphere where new environments have evolved, in geographical time scale, it does not take long for insect species to evolve alongside it and form a symbiotic relationship. In subzero environments many orders of insect families can be seen moving around or may be unseen in a form of dispause beneath the snow. Diapause is defined by Campbell et al. 2002 as a period of suspended development in an insect, an insect equivalent to hibernation so they are not bracing a harsh winter.
Some insects however developed biological mechanisms to withstand subzero temperatures, one of the most common is the development of antifreeze. Antifreeze can allow an insect to stay alive and active during winter but it also allows some insects to freeze solid without dying, the antifreeze controls where the ice forms not allowing it to rupture cells (Burchsted 2009).
Antifreeze proteins, also known as thermal hysteresis proteins are a family which have the ability to inhibit ice growth. Thermal hysteresis is defined by Graether and Sykes as the temperature divergence between the melting point and the non-equilibrium hysteresis freezing point (HFP) of a solution.
Some insects however developed biological mechanisms to withstand subzero temperatures, one of the most common is the development of antifreeze. Antifreeze can allow an insect to stay alive and active during winter but it also allows some insects to freeze solid without dying, the antifreeze controls where the ice forms not allowing it to rupture cells (Burchsted 2009).
Antifreeze proteins, also known as thermal hysteresis proteins are a family which have the ability to inhibit ice growth. Thermal hysteresis is defined by Graether and Sykes as the temperature divergence between the melting point and the non-equilibrium hysteresis freezing point (HFP) of a solution.
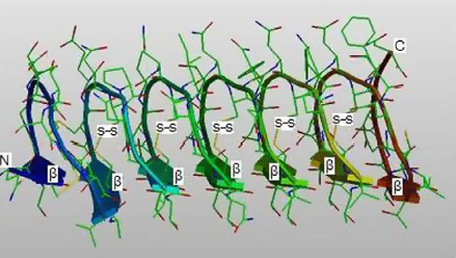
Fig 1. Predicted 3D structure of the Microdera punctipennis antifreeze protein (Qui et al. 2010)
The inhibition of ice growth that the antifreeze proteins cause works by proteins binding to the ice surface in an absorption-inhibition mechanism (Qiu et al. 2010). The absorption-inhibition mechanism is successful as the protein binds to nascent ice nuclei, stopping chemical bonds from forming and halting the change in state of water from liquid to ice. In short an anti freeze has the ability to distinguish the differences between water as a liquid and solid, discriminating between their different structural states (Sharp 2011).
The antifreeze proteins lower the freezing temperature of hemolymph and other fluids without significantly affecting melting points (Qui et al. 2010). An insect can further drop the freezing temperature of its hemolymph by partially dehydrating its cells.
Contrary to popular belief an insect does not have to be in an area dominated by colder months to have developed antifreeze. A research study done by Qui et al. cloned the desert beetle Microdera punctipennis antifreeze protein and heterogeneously expressed it in E. Coli as a fusion protein. The study concluded the structural features of the anti freeze protein account for its stability not only in the cold but extreme heats.
The sequence of Microdera punctipennis antifreeze protein showed tandem repeats of 12-aa with a cysteine in every sixth position. The tertiary structure is a regulated beta helix with the cysteine as the disulfide-bridge allows for a maximum rigid structure and heat stability of the protein thus allowing for its diversification among insects and extreme environments (Qui et al. 2010).
The antifreeze proteins lower the freezing temperature of hemolymph and other fluids without significantly affecting melting points (Qui et al. 2010). An insect can further drop the freezing temperature of its hemolymph by partially dehydrating its cells.
Contrary to popular belief an insect does not have to be in an area dominated by colder months to have developed antifreeze. A research study done by Qui et al. cloned the desert beetle Microdera punctipennis antifreeze protein and heterogeneously expressed it in E. Coli as a fusion protein. The study concluded the structural features of the anti freeze protein account for its stability not only in the cold but extreme heats.
The sequence of Microdera punctipennis antifreeze protein showed tandem repeats of 12-aa with a cysteine in every sixth position. The tertiary structure is a regulated beta helix with the cysteine as the disulfide-bridge allows for a maximum rigid structure and heat stability of the protein thus allowing for its diversification among insects and extreme environments (Qui et al. 2010).

Fig 2. Thermal imaging inside Apis mellifera hive
Homologous antifreeze proteins found in insects that live in diverse environments may be very similar in sequences or protein structure but increasingly different in the temperature dependencies of their activity and stability. Antifreeze proteins with the greatest structural stability are quickly becoming more desirable for uses such as:
Along with physiological and anatomical mechanisms, insects have also developed behavioural adaptations to warm themselves. As the sun rises, many winged insects such as butterflies and bees will bask in the light with their wings open at a 45 degree angle perpendicular to the sun’s rays. Beating their wings slowly in a process known as shivering; bodily fluids are circulated at an accelerated rate through the veins in the wings. While resting their wings at this angle a reflective surface is created, warming the circulating fluid and sending it back into the body resulting in the insect warming.
Colonial insects such as Apis mellifera, the honey bee, stores food in the hive for winter consumption. Honey bees move around their hive in winter beating their wings and warming the hive to stop it from freezing (Figure 2). The honey bees use an exothermic reaction to heat the hive as the food they eat is processed into energy which the bee can use to beat its wings and disperse heat created from the beating of the wings.
- Additive to improve quality of shelf life of frozen products
- Cryoprotective agent
- Cryopreservation
- Cryosurgery
- Genetic splicing to create flora and fauna more tolerable to cold climates.
Along with physiological and anatomical mechanisms, insects have also developed behavioural adaptations to warm themselves. As the sun rises, many winged insects such as butterflies and bees will bask in the light with their wings open at a 45 degree angle perpendicular to the sun’s rays. Beating their wings slowly in a process known as shivering; bodily fluids are circulated at an accelerated rate through the veins in the wings. While resting their wings at this angle a reflective surface is created, warming the circulating fluid and sending it back into the body resulting in the insect warming.
Colonial insects such as Apis mellifera, the honey bee, stores food in the hive for winter consumption. Honey bees move around their hive in winter beating their wings and warming the hive to stop it from freezing (Figure 2). The honey bees use an exothermic reaction to heat the hive as the food they eat is processed into energy which the bee can use to beat its wings and disperse heat created from the beating of the wings.
